Background
Acid-base disorders can indicate potentially life-threatening conditions that require immediate attention. Accurate laboratory assessment and prompt interpretation are central to the evidence-based, cost-effective management of these disorders. A high anion gap metabolic acidosis in the absence of uremia often signals time-sensitive diagnoses such as diabetic ketoacidosis (DKA) or sepsis with lactic acidosis–entities needing immediate and life-saving measures.
Case Presentation
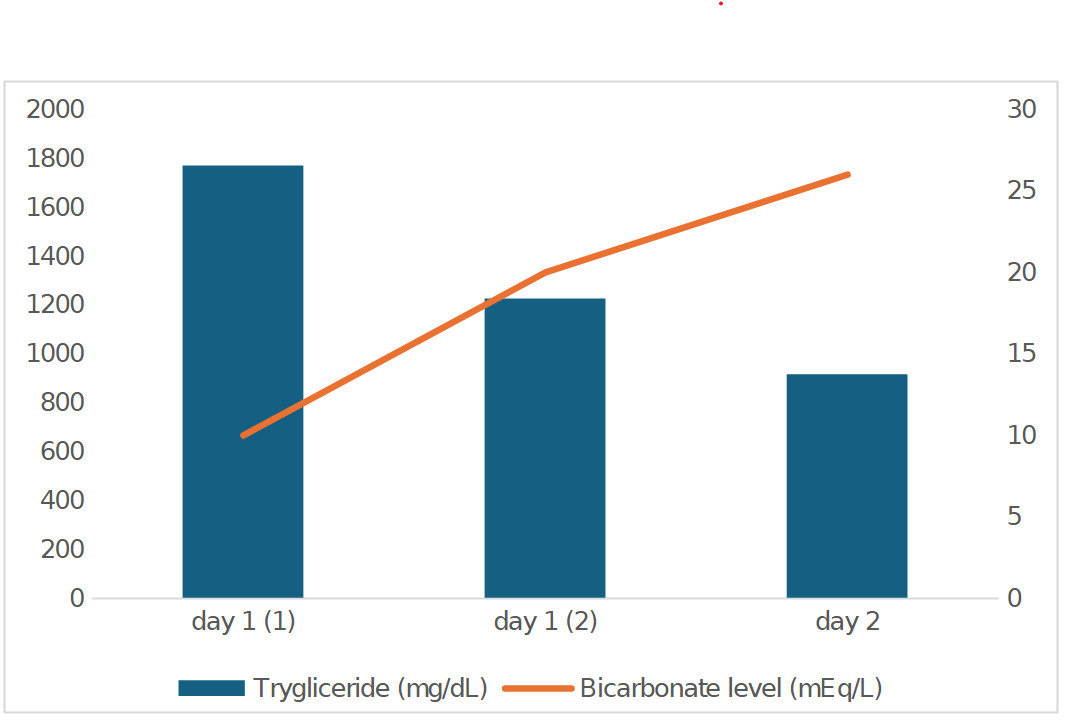
A 30-year-old with no documented medical history presented to the emergency department four days after sustaining a mechanical fall. The initial laboratory workup revealed a significantly elevated anion gap (>25 mEq/L) and extremely low bicarbonate level (<10mEq/L) on his metabolic panel. Due to the abnormal results, these findings were considered erroneous, prompting a repeat laboratory workup that revealed similar findings. Arterial blood gas (ABG) showed a pH of 7.44, PCO2 of 42 mmHg, and bicarbonate level of 28.5 mEq/L. Given the normal blood gas findings, a third blood sample was obtained, which revealed a persistently low bicarbonate level (<10 mEq/L) with an anion gap>23 mEq/L. A bicarbonate drip was started, which was later discontinued when nephrology suspected pseudohypobicarbonatemia. The fasting lipid panel revealed significantly elevated cholesterol of 375 mg/dL and triglycerides of 1769 mg/dL. Imaging studies of the chest and abdomen did not reveal any traumatic injuries and no evidence of acute pancreatitis. He was diagnosed with pseudohypobicarbonatemia and pseudo-anion gap metabolic acidosis secondary to hypertriglyceridemia. Given the lack of pancreatitis, an insulin infusion treatment was deferred, and the patient was started on a fibrate and statin, with strict dietary fat restriction and nutrition counseling. A temporal relationship between the triglyceride levels and measured bicarbonate is shown in Figure 1. Repeat metabolic panel later the same day showed an anion gap of 13 mEq/L with bicarbonate of 20 mEq/L. Lipid panel (non-fasting) showed a total cholesterol of 303 mg/dL with triglycerides of 1224 mg/dL. ABG within 1 hour showed pH of 7.43, PCO2 44 mmHg, and bicarbonate 29.2 mEq/L. On the following day, the anion gap was 7 mEq/L and a bicarbonate of 26 mEq/L, but fasting triglycerides remained elevated at 914 mg/dL. One week after discharge, a repeat metabolic panel showed normal bicarbonate levels, with a non-fasting triglyceride level of 301mg/dL.
Discussion
Bicarbonate is obtained through either direct measurement or a calculated method. Most chemistry analyzers that measure bicarbonate use either an enzymatic/spectrophotometric method or an indirect ion-selective electrode (ISE) method. A calculated bicarbonate value can be obtained from a blood gas sample that directly measures pH and pCO2. The Henderson-Hasselbach equation can be used to estimate bicarbonate concentration. For clinical purposes, calculated and measured bicarbonate are used interchangeably.1 Our laboratory uses the chemistry analyzer Siemens Healthineers Atellica® Solutions CH930 that uses an enzymatic/spectrophotometric method. The blood gas samples are processed on a GEM 5000 analyzer.
Our case revealed an immeasurably low bicarbonate on the basic metabolic panel, but a normal calculated value on the arterial blood gas with a normal pH. When the two values disagree, clinical judgment plays a key role in deciding the next steps. Our patient was clinically stable without concern for life-threatening entities. Suspicion for pseudohypobicarbonatemia (PHB) due to a laboratory interference was raised and the lipid panel revealed severe hypertriglyceridemia with a triglyceride level of 1769 mg/dL, confirming the hypothesis. After starting lipid-lowering therapy, the laboratory interference resolved, revealing a normal bicarbonate, reflecting the underlying acid-base status. Rifkin et al. were the first to hypothesize, in 2014, that the light scattering effect of the lipemic serum drives the laboratory interference when using spectrophotometry, and the interference would not take place with analyzers that use an ISE method, which do not use spectrophotometry.2
Multiple interferences with chemistry analyzers that can lead to pseudohypobicarbonatemia have been described in the literature, including hemolysis, jaundice, paraproteins, and hypertriglyceridemia.3 Determinations of the lipemic index (L-index) or triglyceride concentrations are used to qualify lipemia. The soy-based emulsion Intralipid has been used to simulate lipemia in interference studies.4 Lipemia interferes with the chemistry analyzer by altering light scattering. Sample turbidity is only weakly correlated with triglyceride concentration in patient samples.4 Some simulated samples may not behave the same as native lipemic samples.3 In vivo, patient lipid macromolecular profiles are more complex and varied than those of Intralipid, and hence interference assays of the manufacturer may not be comparable to human blood samples.5
The Siemens Healthineers Atellica® Solutions CH930 has built 7 index values (0-6) that define the approximate concentration ranges for lipemia interference. The Atellica assay Instructions for Use (IFU) provide threshold interference concentrations for Lipemia. Specifically, for bicarbonate, a lipemia concentration of 750 mg/dL of Intralipid would trigger a Lipemia index of 4.6 Even though our sample had a triglyceride level of 1769 mg/dL, it never triggered a lipemia index of more than 3. We hypothesize that, in our case, the inherent variability of the patient’s sample, containing a heterogeneous blend of lipid particles, scattered light differently on separate occasions, leading to a lower lipemia index.
Several methods of removing the lab interference have been described in the literature. Rifkin and Shaub performed a reanalysis of lipemic blood samples after treatment with a lipid-clearing agent, which resulted in a marked improvement in bicarbonate levels.2 Stein completed a preliminary single-institutional retrospective analysis of autoanalyzer-measured versus ABG-calculated bicarbonate values in all patients with triglyceride levels of more than 1000mg/dL chosen in a consecutive fashion over a period of 6 months. The percentage difference between measured and calculated bicarbonate concentrations ranged from 4.5% to 74%, with a mean difference of 29%. He also suggested either ultracentrifuging the lipemic sample or adding a lipid-clearing agent before reporting the results.5 Varghese et al. noted that simple centrifugation does not eradicate the artifact, and that ultracentrifugation may prevent lipid interference, quoting Stein.7 Upon literature review, we could not find a study to suggest a repeated and validated method to manage lipemic samples with pseudohypobicarbonatemia.
In conclusion, clinicians should be aware that hypertriglyceridemia can interfere with chemistry analyzers. When one encounters unexpectedly low bicarbonate values measured by an enzymatic method, one should consider confirming the result with another technique (e.g., an ISE-based analyzer or a blood gas analyzer). Recognizing pseudohypobicarbonatemia may facilitate the timely identification of treatable causes of lipemia or other laboratory interference. Further studies will be needed to assess whether ultracentrifugation is a reliable and reproducible method for eliminating interference in lipemic samples. It should be noted that clinicians should not rely on lipemia flags in isolation, and alternative differentials should be considered based on a complete clinical and diagnostic workup, especially in cases such as ours, where lipemia was not flagged.
Disclosures/Conflicts of Interest
The authors have no conflicts of interest to disclose.
Corresponding author
Ela-Anamaria Moisa, MD
Division of Hospital Medicine
101 Page Street, St Luke’s Hospital,
New Bedford, MA 02740, USA
Email: moisae@southcoast.org