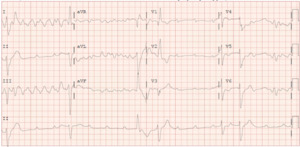
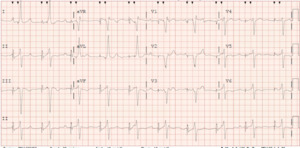
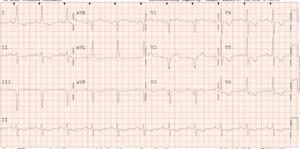
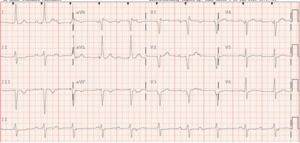
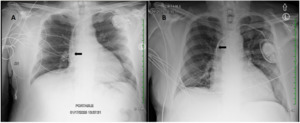
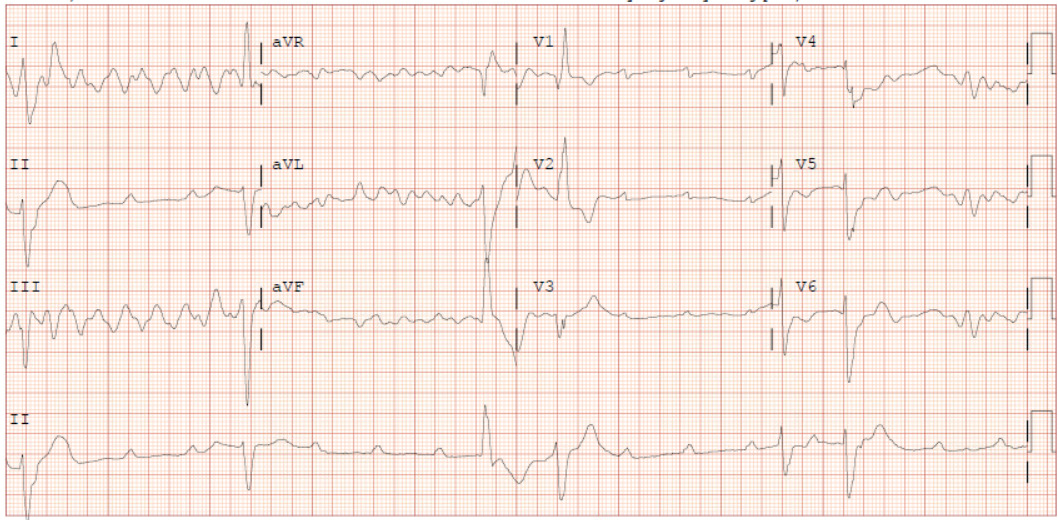
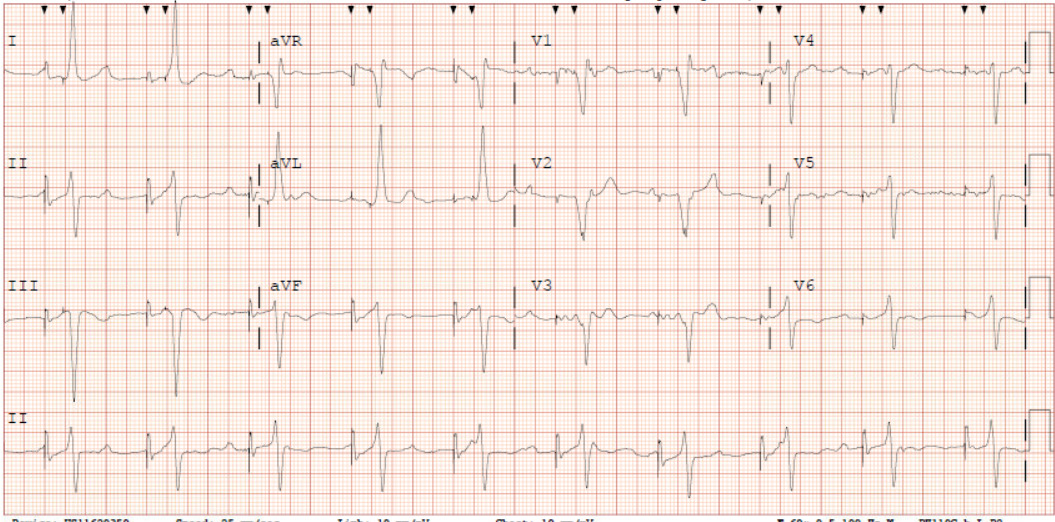
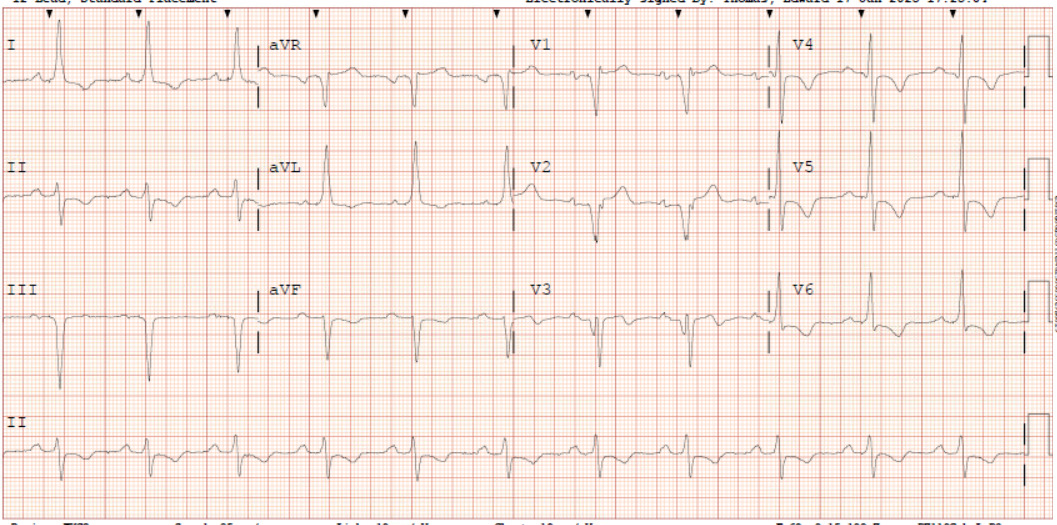
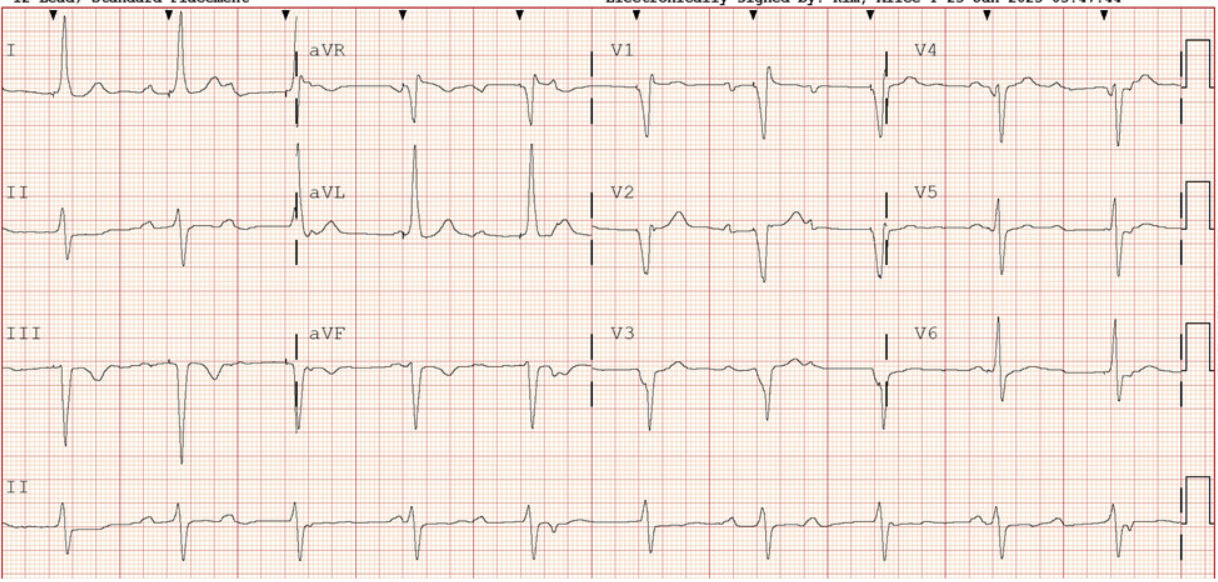
A 62-year-old male with a past medical history notable for left nephrectomy and recent complete heart block status post dual-chamber pacemaker insertion (Figures 1A and 1B) presented with right-sided pectoralis major twitching and persistent singultus. Symptoms began insidiously on post-procedure day five, and after two days of persistent singultus, the patient presented to the hospital. Vital signs were stable, and physical examination revealed ongoing singultus, visible rhythmic contractions of the right chest wall, and no signs of infection at the insertion site or clinical signs of overt heart failure. He denied cough, fever, chest discomfort, shortness of breath, inducible fasciculations, or extremity hypertonia. An electrocardiogram (Figure 1C) demonstrated a paced rhythm consistent with atrioventricular sequential pacing; however, an immediate repeat electrocardiogram (Figure 1D) revealed atrial dissociation with ventricular pacing.
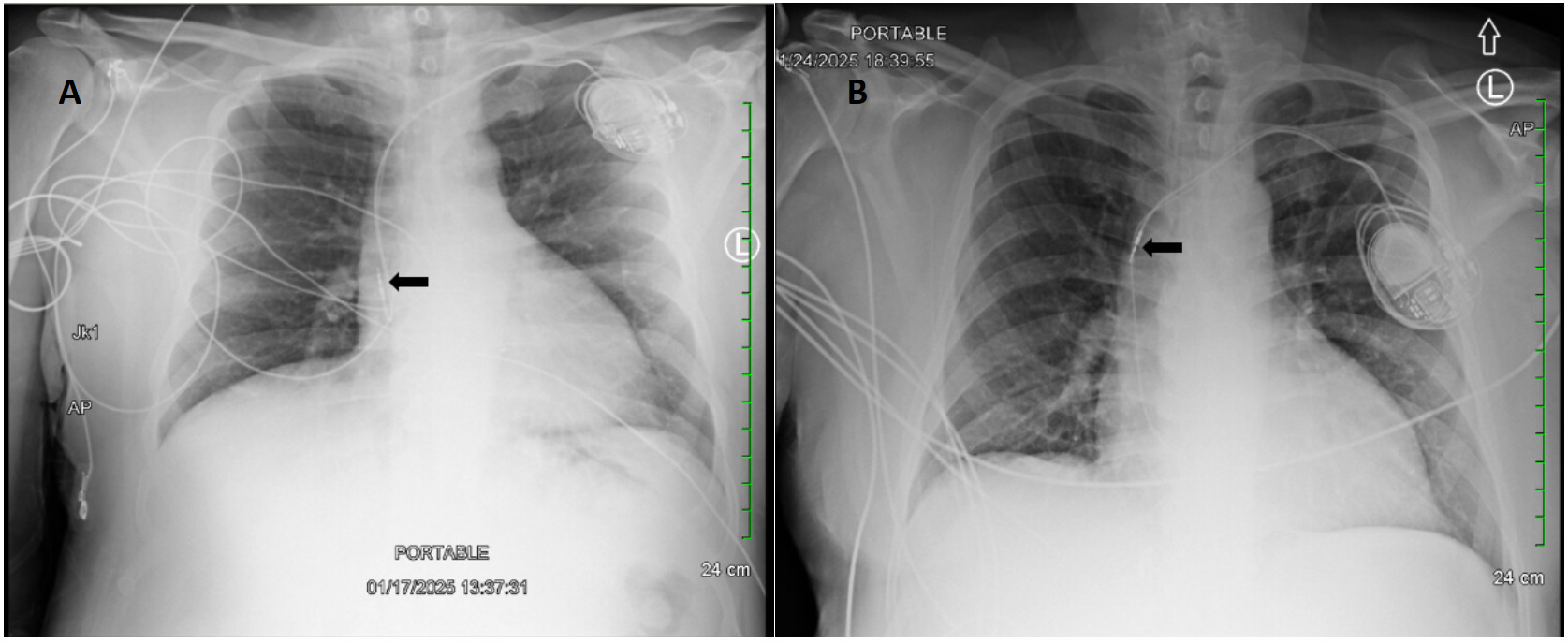
Chest X-rays revealed interval retraction of the atrial lead compared to post-implantation films, with the atrial lead showing migration to the superior vena cava rather than its previous position in the right atrial appendage (Figure 2). Device interrogation confirmed atrial lead dislodgement and subsequent stimulation of the adjacent phrenic nerve. To mitigate symptoms, the pacemaker was reconfigured from initial dual-chamber, rate-adaptive programming (DDDR) to ventricular demand, and rate-adaptive programming (VVIR) to bypass the atrial lead. This change successfully eliminated the diaphragmatic stimulation. He was discharged in stable condition with plans for outpatient atrial lead revision four days later, which was successful.
Cardiac implantable electronic devices are widely used in managing a multitude of arrhythmias and nodal dysfunction.1 While implantation is generally safe and the benefits of performing it typically outweigh the risks, given the usual urgent and emergent indications for an implantable cardiac device, complications can be unavoidable. Complications may include pocket-site infection, hematoma, pneumothorax, and lead dislodgement.2 Of these, lead dislodgement occurs at an estimated incidence of less than 2% and typically within the first 30 days.3 Dislodgement of leads can present in an atypical manner, given the close proximity of neurovascular bundles to implantation.
Phrenic nerve stimulation is a rare presentation that can mimic neuromuscular disorders or diaphragmatic pathology.4 Lead dislodgement is an uncommon yet clinically significant complication of cardiac implantable electronic device placement, most frequently occurring within the first 24–48 hours following implantation.5 The reported prevalence ranges from approximately 1% to 2.7%, and identified patient-related risk factors include female sex and elevated body mass index.6,7 Notably, atrial leads carry a nearly 3.5-fold higher risk of dislodgement than ventricular leads, likely reflecting the thinner atrial wall and greater cardiac motion in the atrium relative to the ventricle.8
This case highlights lead dislodgement as a rare yet clinically significant complication of dual-chamber pacing. Phrenic nerve stimulation typically results from the proximity of atrial or epicardial leads to the nerve, or from incorrect programming that causes far-field capture, leading to diaphragmatic contractions.9 This may manifest as persistent singultus, which can significantly impair quality of life. Pacemaker lead placement is confirmed during the procedure and post-implantation via imaging and electrocardiogram. Leads naturally become securely anchored to cardiac tissue through fibrosis, which typically takes 4-6 weeks.5 Imaging, electrocardiogram interpretation, and device interrogation are essential for accurate diagnosis. Management strategies include lead repositioning, reprogramming, or disabling the affected lead. In our patient, a temporary mode switch to VVIR provided symptomatic relief with planned definitive outpatient lead revision.
Clinicians should maintain a high index of suspicion for lead dislodgement when patients present with atypical symptoms following recent pacemaker insertion. As our patient had confirmed ventricular lead function, he maintained sufficient cardiac output and had no symptoms of his previous cardiac conduction disease. Electrical lead dislodgement can result in phrenic nerve stimulation presenting as singultus.10 Device reprogramming can be an effective but temporary measure, as suboptimal settings that require the pacemaker to compensate for the underlying cause may increase the risk of ventricular arrhythmias, pacemaker syndrome, and atrial fibrillation.11 Correction of electrical lead positioning can be performed via additional procedures, including explant and implantation of new leads, percutaneous or surgical repositioning, or abandonment of leads entirely.12 Early recognition of lead dislodgement and its sequelae can prevent unnecessary testing and direct appropriate care.
Conflicts of Interest/Disclosures
The authors have no conflicts of interest to disclose.
Corresponding author
Jinen Thakkar MD, FACP
Department of General Internal Medicine, Brown University
Kent Hospital, RI, USA
Email: jinen_thakkar@brown.edu